
GDT-001 (IL12-FHAB)
Targeted Immune Activation Cancer Therapy,
Turning 'Cold' Tumors 'Hot'
GDT-001 (Previously known as SON - 1010 ) is a candidate immunotherapeutic recombinant drug that links unmodified single-chain human IL-12 with the albumin-binding domain of the single-chain antibody fragment A10m3.
This was selected to bind the patient’s circulating albumin in the body, both at normal pH, as well as at the acidic pH typically found in the Tumor Microenvironment (TME)
The FHAB technology targets tumor and lymphatic tissue, and this Navigated Delivery™ technology has demonstrated significant clinical benefit in clinical studies with sarcoma patients, without any signs of cytokine release syndrome or any adverse side effects. This validating asset will be further expanded through partnerships, while the learning from this clinical experience will help accelerate the clinical development for the exciting dual cytokine assets in the pipeline.

Key Details
Indications
Soft-Tissue Sarcomas (STS) in combination with trabectedin (Yondelis®)
Current Stage
Phase 1
Mechanism of actions
Interleukin-12 can orchestrate a robust immune response to many cancers and pathogens. Given specific proteins induced in the TME that actively accumulate, retain and bind albumin, such as the Secreted Protein and Rich in Cysteine (SPARC) and glycoprotein 60 (GP60), cancers such as non-small cell lung cancer, melanoma, head and neck cancer, sarcoma, and some gynecological cancers are particularly relevant for this approach.
GDT-001 is designed to deliver IL-12 to local tumor tissue, turning 'cold' tumors 'hot' by stimulating IFNγ, which activates innate and adaptive immune cell responses and increases the production of Programed Death Ligand 1 (PD-L1) on tumor cells.
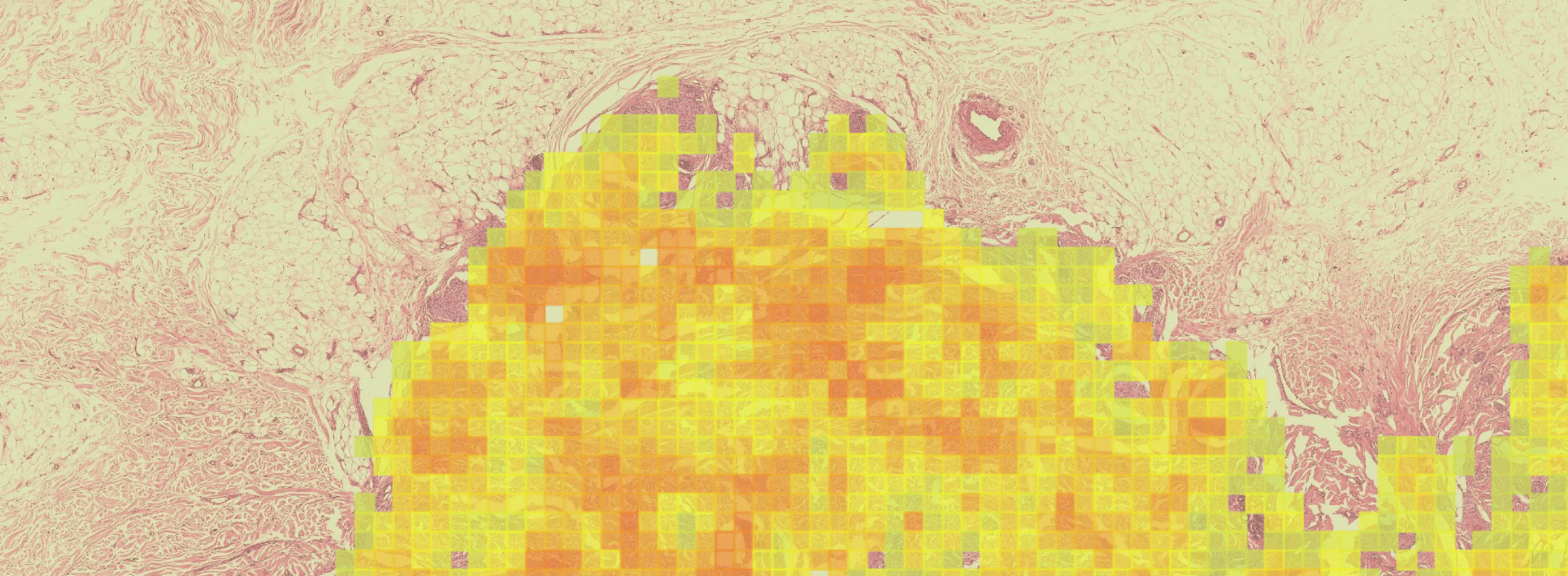
Key Advantages
Demonstrated to Reduce Tumor Growth
GDT-001 is approximately 35-fold more potent than IL-12 alone at Day 10, using a 1:30 ratio of IL-12, demonstrated in Mouse B16F10 Melanoma Model.
Targeted Delivery
The FHAB technology targets tumor and lymphatic tissue, providing a mechanism for dose sparing and an improved safety and efficacy profile.
Related Programs

IND ready | Bladder Cancer and Solid Tumors
GDT-002 (IL12-FHAB-IL15)
A first-in-class IL-12+IL-15 immunotherapy candidate delivering dual cytokine action directly to the tumor microenvironment.

Preclinical | Solid Tumors
GDT-003 (IL- 18BPR
-FHAB-IL12)
A first-in-class IL-12+IL-18 immunotherapy candidate with issued intellectual property delivering dual cytokine action directly to the tumor microenvironment.

Interested in GDT-001 ?
Guidant BioTherapeutics is actively engaging with potential partners and licensees interested in leveraging the strength of our proprietary FHAB technology platform.

