
GDT-002 (IL12-FHAB-IL15)
GDT-002 (IL12-FHAB-IL15)
Advancing for treatment of non muscle invasive bladder cancer (NMIBC) and other solid tumors
Advancing for treatment of non muscle invasive bladder cancer (NMIBC) and other solid tumors
Home > PIPELINE > GDT-002 (IL12-FHAB-IL15)
Home > PIPELINE > GDT-002 (IL12-FHAB-IL15)

Guidant BioTherapeutics
Guidant BioTherapeutics
GDT-002 is an immunotherapeutic bifunctional drug candidate that links unmodified single-chain human IL-12 and human IL-15 with the albumin-binding domain of the single-chain antibody fragment FHAB, separating the two cytokines with linkers to avoid steric hindrance.
The FHAB single chain was selected to bind well at normal pH, as well as at an acidic pH that is typically found in the tumor microenvironment (TME). The FHAB technology targets tumor and lymphatic tissue, providing lowered dose, enhanced PK, and an opportunity to improve the safety and efficacy profile of not only IL-12 and IL-15, but a variety of other potent immunomodulators using the platform.
GDT-002 is an immunotherapeutic bifunctional drug candidate that links unmodified single-chain human IL-12 and human IL-15 with the albumin-binding domain of the single-chain antibody fragment FHAB, separating the two cytokines with linkers to avoid steric hindrance.
The FHAB single chain was selected to bind well at normal pH, as well as at an acidic pH that is typically found in the tumor microenvironment (TME). The FHAB technology targets tumor and lymphatic tissue, providing lowered dose, enhanced PK, and an opportunity to improve the safety and efficacy profile of not only IL-12 and IL-15, but a variety of other potent immunomodulators using the platform.
Key Details
Key Details
OVERVIEW
OVERVIEW
Indications
Indications
Current Stage
Current Stage
Market Opportunity
Market Opportunity
Bladder Cancer and other solid tumors
Bladder Cancer and other solid tumors
IND-ready and starting Phase I
IND-ready and starting Phase I
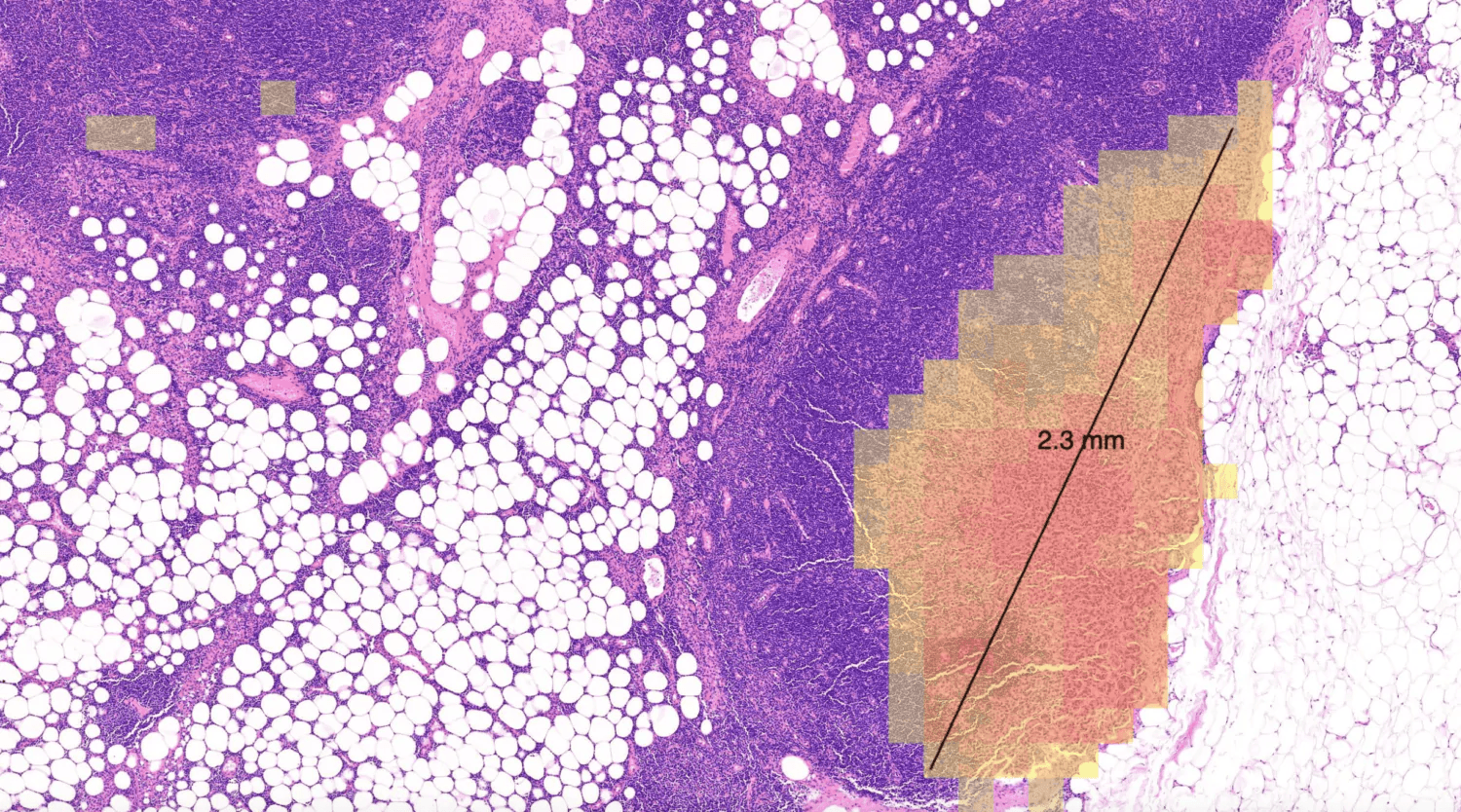
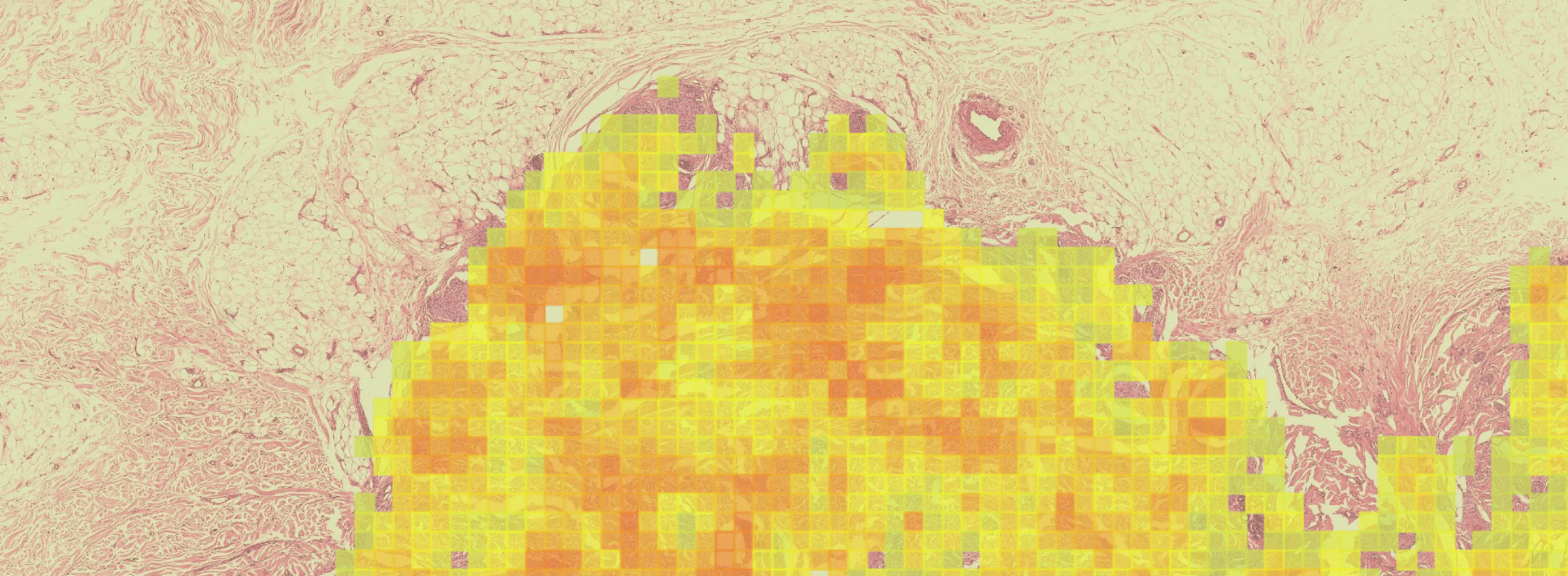
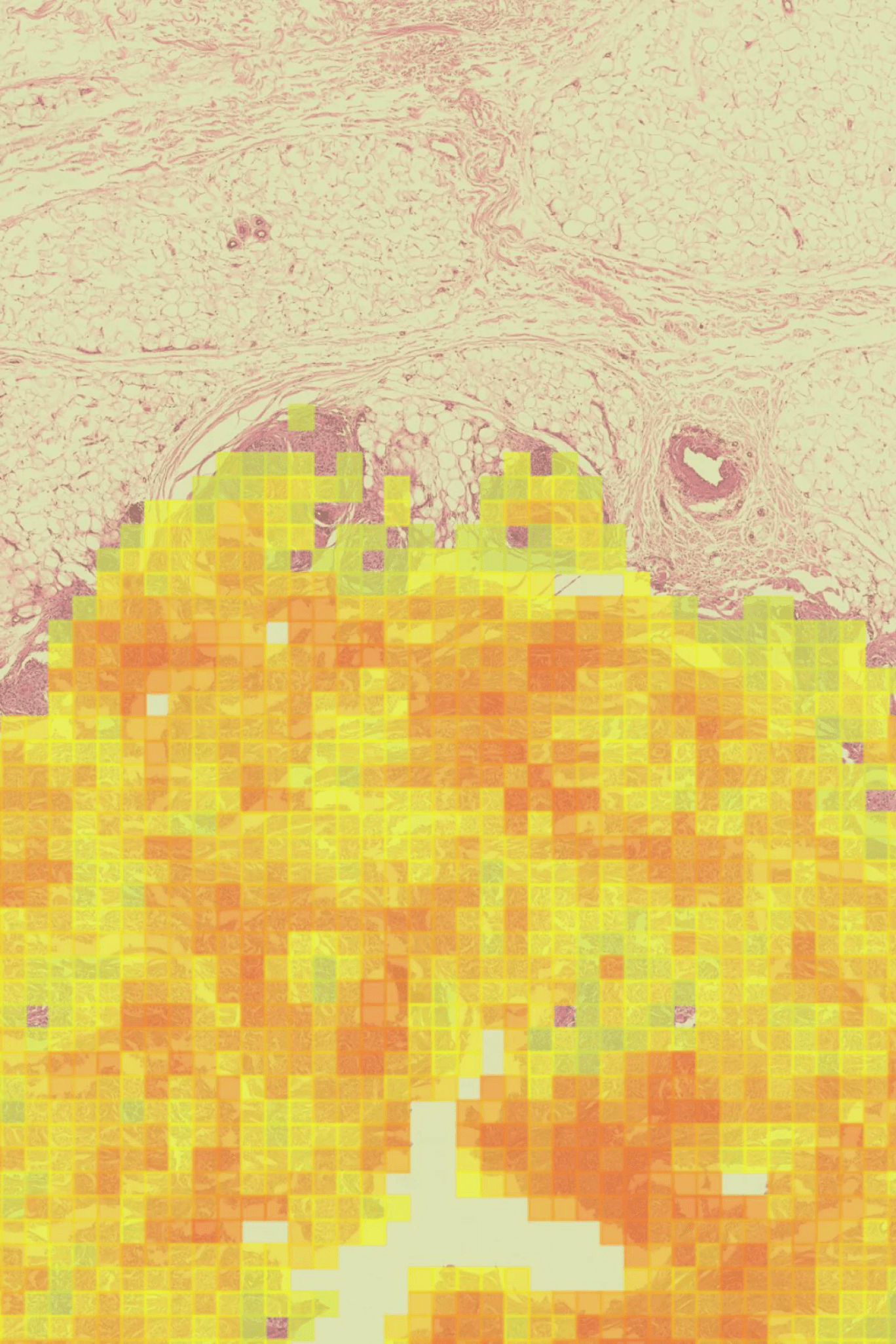
IL-15 alone, co-dosed with BCG vaccine, has proven very effective in non-muscle-invasive bladder cancer. A dual cytokine molecule with navigated delivery (™) that is accumulated and retained in the tumor microenvironment has the potential to also treat advanced and metastatic disease, with lower and less frequent dosing, improving potency with fewer side effects.
IL-15 alone, co-dosed with BCG vaccine, has proven very effective in non-muscle-invasive bladder cancer. A dual cytokine molecule with navigated delivery (™) that is accumulated and retained in the tumor microenvironment has the potential to also treat advanced and metastatic disease, with lower and less frequent dosing, improving potency with fewer side effects.
We believe these dual-targeting cytokines can orchestrate a robust immune response to many cancers and pathogens, particularly when presented together on the same molecule. Given specific proteins induced in the TME that actively accumulate, retain and bind albumin, such as the Secreted Protein and Rich in Cysteine (SPARC) and glycoprotein 60 (GP60), cancers such as bladder cancer, non-small cell lung cancer, melanoma, head and neck cancer, sarcoma, and some gynecological cancers are particularly relevant for this approach.
GDT-002 is designed to navigate IL-12 and IL-15 to accumulate in local tumor tissue, with the intention of turning 'cold' tumors 'hot' by stimulating IFNγ, which activates both innate and adaptive immune cells in the TME, as well as increasing the production of Programed Death Ligand 1 (PD-L1) on tumor cells.
We believe these dual-targeting cytokines can orchestrate a robust immune response to many cancers and pathogens, particularly when presented together on the same molecule. Given specific proteins induced in the TME that actively accumulate, retain and bind albumin, such as the Secreted Protein and Rich in Cysteine (SPARC) and glycoprotein 60 (GP60), cancers such as bladder cancer, non-small cell lung cancer, melanoma, head and neck cancer, sarcoma, and some gynecological cancers are particularly relevant for this approach.
GDT-002 is designed to navigate IL-12 and IL-15 to accumulate in local tumor tissue, with the intention of turning 'cold' tumors 'hot' by stimulating IFNγ, which activates both innate and adaptive immune cells in the TME, as well as increasing the production of Programed Death Ligand 1 (PD-L1) on tumor cells.
Mechanism of actions
Mechanism of actions
Bifunctional Action
Bifunctional Action
GDT-002 links IL-12 and IL-15 on the same molecule, separated by linkers to avoid steric hindrance, designed to work together to generate a robust immune response in the tumor microenvironment.
GDT-002 links IL-12 and IL-15 on the same molecule, separated by linkers to avoid steric hindrance, designed to work together to generate a robust immune response in the tumor microenvironment.
Dose-Sparing and Enhanced PK
Dose-Sparing and Enhanced PK
Advantage one
Advantage one
Advantage two
Advantage two
The FHAB technology provides a mechanism for dose-sparing and enhanced pharmacokinetics, improving the safety and efficacy profile of both IL-12 and IL-15.
The FHAB technology provides a mechanism for dose-sparing and enhanced pharmacokinetics, improving the safety and efficacy profile of both IL-12 and IL-15.
Key advantages
Key advantages
Related Programs
Related Programs
First in class dual-cytokine IL-12 + IL-15 candidate ready for IND and clinical trials.
First in class dual-cytokine IL-12 + IL-15 candidate ready for IND and clinical trials.
GDT-001 (IL12-FHAB)
GDT-001 (IL12-FHAB)

Read More
Read More
Phase 1 | Solid Tumors
Phase 1 | Solid Tumors
First in class dual-cytokine IL-12 + IL-18 candidate in early preclinical development.
First in class dual-cytokine IL-12 + IL-18 candidate in early preclinical development.
GDT-003 (IL12-FHAB-IL18)
GDT-003 (IL12-FHAB-IL18)

Read More
Read More
Phase 1 | Solid Tumors
Phase 1 | Solid Tumors

Interested in GDT-001?
Interested in GDT-001?
Partnerships Inquiry
Partnerships Inquiry
Guidant BioTherapeutics is actively engaging with potential partners and licensees interested in leveraging the strength of our proprietary FHAB technology platform.
Guidant BioTherapeutics is actively engaging with potential partners and licensees interested in leveraging the strength of our proprietary FHAB technology platform.
Partner With Us
Partner With Us

Guidant BioTherapeutics
Guidant BioTherapeutics
© 2026 Sonnet BioTherapeutics, Inc. All Rights Reserved.
© 2026 Sonnet BioTherapeutics, Inc. All Rights Reserved.
CONTACT
CONTACT
newsletter
newsletter

GDT-002 (IL12-FHAB-IL15)
Advancing for treatment of non muscle invasive bladder cancer (NMIBC) and other solid tumors
Home > PIPELINE > GDT-002 (IL12-FHAB-IL15)

Guidant Biotherapeutics
GET IN TOUCH
GDT-002 is an immunotherapeutic bifunctional drug candidate that links unmodified single-chain human IL-12 and human IL-15 with the albumin-binding domain of the single-chain antibody fragment FHAB, separating the two cytokines with linkers to avoid steric hindrance.
The FHAB single chain was selected to bind well at normal pH, as well as at an acidic pH that is typically found in the tumor microenvironment (TME). The FHAB technology targets tumor and lymphatic tissue, providing lowered dose, enhanced PK, and an opportunity to improve the safety and efficacy profile of not only IL-12 and IL-15, but a variety of other potent immunomodulators using the platform.

Key Details
OVERVIEW
Mechanism of actions
Related Programs
Indications
Current Stage
Market Opportunity
Bladder Cancer and other solid tumors
IND-ready and starting Phase I
IND-ready and starting Phase I
We believe these dual-targeting cytokines can orchestrate a robust immune response to many cancers and pathogens, particularly when presented together on the same molecule. Given specific proteins induced in the TME that actively accumulate, retain and bind albumin, such as the Secreted Protein and Rich in Cysteine (SPARC) and glycoprotein 60 (GP60), cancers such as bladder cancer, non-small cell lung cancer, melanoma, head and neck cancer, sarcoma, and some gynecological cancers are particularly relevant for this approach.
GDT-002 is designed to navigate IL-12 and IL-15 to accumulate in local tumor tissue, with the intention of turning 'cold' tumors 'hot' by stimulating IFNγ, which activates both innate and adaptive immune cells in the TME, as well as increasing the production of Programed Death Ligand 1 (PD-L1) on tumor cells.
Key advantages
Dose-Sparing and Enhanced PK
Advantage one
The FHAB technology provides a mechanism for dose-sparing and enhanced pharmacokinetics, improving the safety and efficacy profile of both IL-12 and IL-15.
Bifunctional Action
Advantage two
GDT-002 links IL-12 and IL-15 on the same molecule, separated by linkers to avoid steric hindrance, designed to work together to generate a robust immune response in the tumor microenvironment.
First in class dual-cytokine IL-12 + IL-15 candidate ready for IND and clinical trials.
GDT-001 (IL12-FHAB)

Read More
Phase 1 | Solid Tumors
First in class dual-cytokine IL-12 + IL-18 candidate in early preclinical development.
GDT-003 (IL12-FHAB-IL18)

Read More
Phase 1 | Solid Tumors

Interested in GDT-001?
Guidant BioTherapeutics is actively engaging with potential partners and licensees interested in leveraging the strength of our proprietary FHAB technology platform.
Partner With Us
Partnerships Inquiry